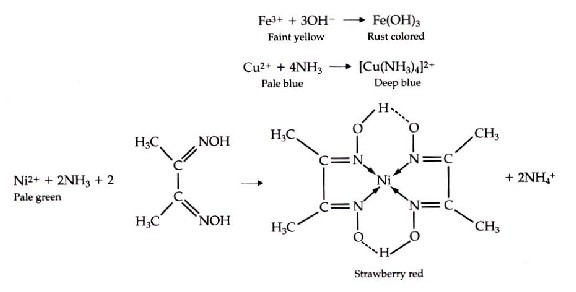
4 Analytical Chemistry 2.1 dimethylglyoxime The discovery, in 1905, that dimethylglyoxime (dmg) selectively precip - itates Ni 2+ and Pd2+ led to an improved analytical method for the quantita - tive analysis of nickel.4 The resulting analysis, which is outlined in Figure 1.2, requires fewer manipulations and less time. Balance the reaction of HDMG + Ni(NO3)2 = Ni(DMG)2 + HNO3 using this chemical equation balancer! The hybridization of Ni in Ni(DMG)2 is dsp2. Ni forms octahedral, square planar and tetrahedral complexes in +2 oxidation state. It acquires stability through chelation and intramolecular hydrogen bonding. In Ni(DMG)2 the nickel is in the +2 oxidation state and to have a square planar geometry because of chelation the pairing of electrons takes place. Nickel-dimethylglyoxime complex (abbreviated as Ni(II)(DMG) 2) modified carbon paste and graphite electrodes were prepared by mixing Ni(II)(DMG) 2 with graphite paste, and coating Ni(II)(DMG) 2 to the graphite surface. It is necessary to cycle the electrode potential to a high value (e.g. 0.8 V versus SCE) for the preparation of the modified electrodes. The hybridization of Ni in Ni(DMG)2 is dsp2. Ni forms octahedral, square planar and tetrahedral complexes in +2 oxidation state. It acquires stability through chelation and intramolecular hydrogen bonding. In Ni(DMG)2 the nickel is in the +2 oxidation state and to have a square planar geometry because of chelation the pairing of electrons takes place. The LibreTexts libraries are Powered by MindTouch ® and are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. We also acknowledge previous National Science Foundation support under grant numbers 1246120, 1525057, and 1413739.
(Redirected from Ni(dmgH)2)
![[ni(dmg)2]2+ [ni(dmg)2]2+](https://www.actugaming.net/wp-content/uploads/2018/03/Ni-no-Kuni-II-1.jpg)
| Names | |
|---|---|
| IUPAC name nickel;N-[(Z)-3-nitrosobut-2-en-2-yl]hydroxylamine | |
| Other names | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
| EC Number | |
PubChemCID | |
| |
| |
| Properties | |
| C8H14N4NiO4 | |
| Molar mass | 288.917 g·mol−1 |
| Appearance | red solid |
| Density | 1.698 g/cm3 |
| Hazards | |
| GHS pictograms | |
| GHS Signal word | Warning |
| H315, H317, H319, H335, H351 | |
| P201, P202, P261, P264, P271, P272, P280, P281, P302+352, P304+340, P305+351+338, P308+313, P312, P321, P332+313, P333+313, P337+313, P362, P363, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Ni(dmg)2 2 0
Nickel bis(dimethylglyoximate) is the coordination complex with the formula Ni[ONC(CH3)C(CH3)NOH]2. The compound is a bright red solid. It achieved prominence for its use in the qualitative analysis of nickel.[1]
Structure[edit]
Nickel(II) is square planar.[2] It is surrounded by two equivalents of the conjugate base (dmgH−) of dimethylglyoxime (dmgH2). The pair of organic ligands are joined through hydrogen bonds to give a macrocyclic ligand. The complex is distinctively colored and insoluble leading to its use as a chelating agent in the gravimetric analysis of nickel.
The use of dimethylglyoxime as a reagent to detect nickel was reported by L. A. Chugaev in 1905.[3]
References[edit]
- ^Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN978-0-08-037941-8.
- ^Donald E. Williams, Gabriele Wohlauer, R. E. Rundle (1959). 'Crystal Structures of Nickel and Palladium Dimethylglyoximes'. J. Am. Chem. Soc. 81: 755–756. doi:10.1021/ja01512a066.CS1 maint: uses authors parameter (link)
- ^Lev Tschugaeff (1905). 'Über ein neues, empfindliches Reagens auf Nickel'. Berichte der deutschen chemischen Gesellschaft. 38 (3): 2520–2522. doi:10.1002/cber.19050380317.
Ni So4 +dmg Ni Dmg 2 2+ Aq
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Nickel_bis(dimethylglyoximate)&oldid=879626005'
